Nuper, Nanjing Limingbio scriptor Novel Coronavirus (Sars, Cov-II) antigen Deprehensio Reagent "Strikstep® Sars, Cov- II-II Antigen Celeros Test" est adeptus est per perficientur verificationem de Paulo-Ehrlich-Institut (Pei *) in Germania, in Germania, in Germany (Pei) in Germania, in Germania est (Pei) in Germania, quod est in Germania (Pei) in Germania, in Germania est (Pei) in Germania, in Germania, in Germania) in Germania Certified by Germanica Foederatum Agency ad medicamenta et medicinae fabrica administratione (BFARM). Limingbio est unum ex paucis manufacturers in Sina quae adeptus est dual certification of BFARM + Pei in Germania. In liming bio est Antigen celeri test transiit auctoritate certification de ministerium de salute multa regionibus, quae plene probat optimum perficientur ad ornamentum.


Liming Bio est Antigen celeri test feliciter Transierunt Germanico Pei perficientur verificationem
Ps Pei: Paulus Ehrlich Institutum (German: Paulus-Ehrlich-Institut), etiam notum est quod Germanico Foederatum Institute of Vaccines et Biomedicine est a Germanica Foederationis et Medical Ministerio de Salutem (BMG ), Habet independens perficientur biologicum productum inspectionem, orci iudicium probat, productum probat et ipsum, et batch issuance. Simul etiam suscipit didicit, revision pertinet ordinationes etprovidosScientific pro diversis organizationibus, praesertim aliquam Europaeam unionem regionibus Europaeae unio et internationalis committees. ALSO,providosProfessional consilium ad German Imperium, loci agencies et parliamentum, et providerespertinet notitia ad aegris et consumers.
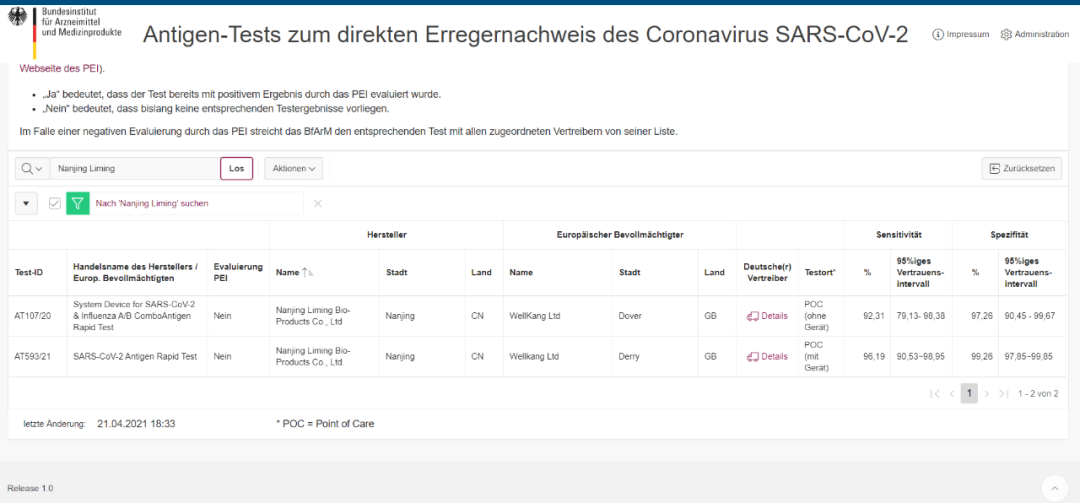
Liming Bio est Antigen celeri test feliciter Transierunt German BFARM Certification
Et StriksEp® Sars, Cov-II Antigen celeri Test developed by Nanjing Liming Bio habet successive adeptus Europaeae Unionis CE Certification, in Sinis National Institutum pro cibo et medicamento, intravit in Rockefeller Foundation Suspendisse List, et Guatemala Certification, Italian Ministerio Salutem Certification, German Certification, Aequatoria Certification, Brazil (Anvisa) Certification, Argentina Certification, Argentina, Singapore HSA Certification, Mexica Certification, Indonesia Certification, Thailand Certification. Hoc vicit laudem in independens iudicium de Britannia Department of Health et Humanum Services (Dhsc) et (Britanniae AAA Certification).

Malaysia MDA Novel coronavirus antigen sui-test certificatorium
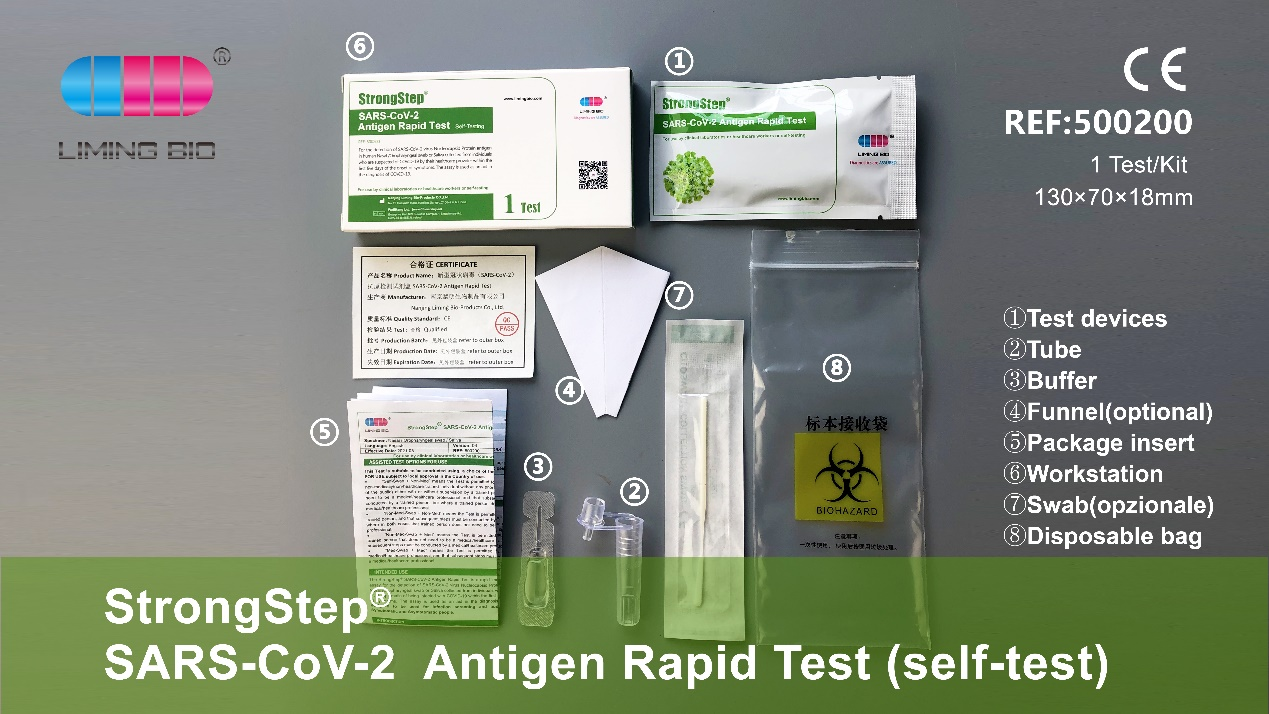
Features et commoda
I convenient sampling: Non-Psidium sample Collection, saliva et nasopharyngeal swab.
II ieiunium deprehendatur: et totius deprehendatur processus tantum accipit XV minuta, et eventus sunt directe observantur a oculis.
III Simple operatio: quod potest esse operated sine ulla auxilia apparatu et absque ulla experientia.
IV optimum perficientur: specificitas est 99,26%, sensus est 96.2% et altiore accurate est XCV%.
V demanda Aliquam, ad praesens, in comitatu habet professional medical version, domum auto-test (saliva + nasopharyngeal swab) version et mini sui test version, etc. Packaging arca et instructiones potest esse customized secundum elit.
This System Device for SARS-CoV-2 Antigen Rapid Test (pen type) is equipped with a biological safety protection device, which can effectively block the virus in the specimen processing solution from volatilizing into the air, polluting the environment, and effectively protect the Operator in Deprehensio Sars-Cov- II antigen celeri test.
In current global pestilentia situ adhuc gravibus. Cum emergentiæ et propagationem de variants of novae coronavirus virus, in multis regionibus et regiones resilientem, et praeventionis et control nisus sunt adversus maiorem provocationes. Et Sars-Cov-II antigen celeri test est ieiunium, accurate, simplex ad operari, et requirit humilis apparatu et personas. Est idoneam ad celeri inquisitionis suspected casibus magnarum scala novus coronam virus infectio, et maxime efficax ad celeri diagnosi concentrata motum. Potest adhiberi sicut prima linea defensionis ad pestilentiam potestate, applicantur ad deprehendatur mane infectiones, ut adiuvaret epidemiam praeventionis et imperium, et control propagationem de virum.
Nanjing Liming Bio-Products Co., Ltd. condita est in MMI. Est a biologicum diagnostic turba specialiter in investigationis et progressionem, productio et venditionesque in orci microbial in vitro Herpesvirus. Habet XX annos optimum qualis et exaggeratus est completum qualis ratio, et adeptus est is013485 certification. Productio administratione currit proprie secundum internationalis qualitas administratione ratio, cursus, quod summus qualitas products serve customers totus super orbis terrarum. Is est paulatim developed in an internationally clarus magna et medium-amplitudo summus tech coeptis specializing in R & D, productio, Sales et servitium in Vitro celeri diagnostic reagents.
Post tempus: Oct-28-2021







